Clinical Trial Imaging:
A Complete Guide
Clinical trial imaging involves collecting, transferring, reviewing, and analyzing medical images such as CT, MRI, PET, and ultrasound scans across multiple investigator sites. Managing this process efficiently is critical for reliable study endpoints, regulatory compliance, and timely trial execution.
Medical imaging plays a central role in modern clinical trials. Imaging techniques such as CT, MRI, PET, and ultrasound allow researchers to measure disease progression, evaluate treatment response, and monitor safety outcomes.
Because imaging data is complex and sensitive, clinical trials require structured workflows for image collection, quality control, analysis, and review. These workflows ensure that imaging endpoints are reliable and consistent across sites and readers.
This guide explains how imaging is used in clinical trials and how imaging data is managed throughout the research process.
What Is Clinical Trial Imaging?
Clinical trial imaging refers to the use of medical imaging modalities to support clinical research studies. Imaging data may be used to:
-
assess treatment response
-
measure tumor size
-
evaluate structural changes
-
detect disease progression
-
monitor safety outcomes
Common imaging modalities used in clinical trials include:
-
CT (Computed Tomography)
-
MRI (Magnetic Resonance Imaging)
-
PET (Positron Emission Tomography)
-
Ultrasound
-
X-ray
These images are collected at investigator sites and transferred to research systems where they can be reviewed by trained readers.
Why Imaging Is Important in Clinical Trials
Imaging provides objective measurements that complement clinical and laboratory endpoints.
For example, oncology trials frequently use imaging to evaluate tumor response to therapy. Neurology trials may use MRI to measure structural brain changes. Musculoskeletal studies may use ultrasound or MRI to evaluate joint inflammation.
Imaging endpoints allow researchers to track disease progression with precision and reproducibility.
However, imaging introduces operational challenges. Clinical trials may involve hundreds of investigator sites across multiple countries, each producing imaging data that must be standardized and reviewed.
Imaging Workflow in Clinical Trials
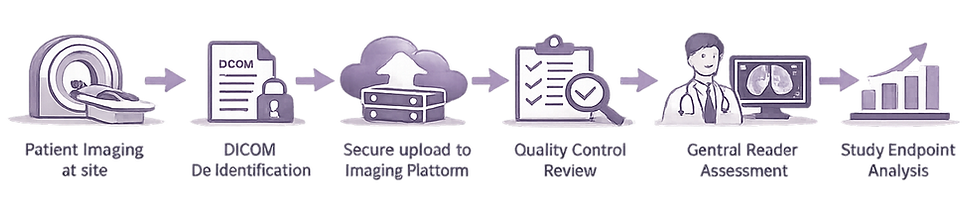
A typical imaging workflow in a clinical trial involves several steps.
-
Image acquisition at the investigator site: Patients undergo imaging procedures according to the study protocol. Imaging parameters must follow predefined protocols to ensure consistency across sites.
-
Image preparation and de-identification: Before images can be transferred to research systems, patient identifiers must be removed. This process is called DICOM de-identification. Patient privacy regulations such as HIPAA and GDPR require that personal information is removed before images are shared.
-
Image upload and storage: De-identified images are uploaded to secure research platforms where they are stored and organized according to patient, visit, and study.
-
Quality control (QC): Quality control teams review images to ensure they meet protocol requirements. QC checks may include: correct imaging modality, correct anatomical coverage, acceptable image quality, protocol compliance.
-
Central imaging review: Independent readers evaluate the images according to predefined criteria. For example, oncology trials often use RECIST criteria to assess tumor response.
-
Data analysis and reporting: Results from image assessments are analyzed and reported to support study endpoints.
Challenges in Clinical Trial Imaging
Managing imaging data across multiple sites introduces several challenges.
-
Standardization: Sites may use different scanners and protocols, which can introduce variability in imaging results.
-
Data privacy: Medical images contain sensitive patient information that must be protected during transfer and storage.
-
Data volume: Imaging studies generate large volumes of data that must be securely stored and organized.
-
Reader consistency: Central readers must follow standardized criteria to ensure consistent assessments across patients and timepoints.
Quality Control in Imaging Trials
Quality control is essential for ensuring reliable imaging data. QC teams typically verify:
-
protocol compliance
-
correct patient and visit assignment
-
absence of identifying information
-
acceptable image quality
Poor quality images can lead to unreliable measurements and may require repeat imaging.
Central Imaging Review
Many clinical trials use central reading to evaluate imaging endpoints.
In central review workflows:
-
images are reviewed by independent readers
-
readers follow standardized scoring criteria
-
discrepancies may trigger adjudication
Central review reduces variability and ensures consistent evaluation across sites.
Imaging Endpoints in Clinical Trials
Imaging endpoints are measurements derived from medical images that support study objectives. Examples include:
-
tumor size measurements
-
lesion counts
-
structural changes in organs
-
quantitative imaging biomarkers
In oncology trials, RECIST (Response Evaluation Criteria in Solid Tumors) is commonly used to measure tumor response.
Other studies may use specialized imaging scoring systems depending on the disease area.
The Role of Imaging Platforms
Because imaging workflows involve multiple stakeholders, many studies use specialized imaging platforms to manage data.
These platforms typically provide:
-
secure image upload
-
automated de-identification
-
workflow management
-
quality control tools
-
centralized image viewing
-
reader reporting tools
Cloud-based platforms allow investigators, QC teams, and readers to collaborate across geographic locations.
How SliceVault Supports Clinical Trial Imaging
SliceVault is a cloud-based platform designed to support imaging workflows in clinical trials. Key capabilities include:
-
secure upload of imaging data
-
automatic DICOM de-identification
-
integrated image viewer
-
workflow management for QC and readers
-
structured reporting tools
By centralizing imaging workflows, research teams can streamline data management and ensure consistent evaluation across sites.
Conclusion
Imaging plays a vital role in clinical trials by providing objective measurements of disease progression and treatment response.
However, managing imaging data requires structured workflows for acquisition, de-identification, quality control, and review.
Modern imaging platforms help research teams manage these processes efficiently while maintaining compliance with privacy regulations and study protocols.
As imaging endpoints become increasingly important in clinical research, robust imaging data management systems will continue to play a critical role in successful trials.